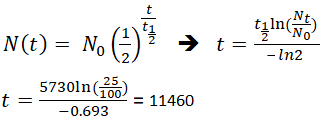
is the time in the same units as the half-life, is the half-life.
Half life formula math free#
"Radiogenic Isotope Geology." United Kingdom: University Press, Cambridge. Effective half-life - Wikipedia, the free encyclopedia Radioactive decay - Wikipedia, the free encyclopedia The equation you want is (equation 1) where is the original amount of material present. KAERI Chart of the Nuclides has lots of half-lives you can look up.The Radioactive Decay page has a much more detailed and thorough description of radioactive decay, including situations with more complex decay chains.There are many steps along the way, but they all have very short half-lives compared to that of U-238 so they can be ignored on these long (geologic) time scales. The decay of Uranium-238 eventually leads to Lead-206. The relationship between the half-life, T1/2.

Notice that the sum of these two curves is always 1.0.įigure 2. The time required for half of the original population of radioactive atoms to decay is called the half-life. $$-\frac N\\Īn example of Uranium-238 (with a 4.47 billion year half-life) decaying into Lead-206 is shown in Figure 2, where we assume \(D_0 = 0\). If you had 10 jumping beans and saw one jump every second, you’d expect to see about 10 jumps per second if you had 100. Decay Formula Formula for Half-Life in Exponential Decay. The basic assumption of radioactive decay is that the amount y of 14C in the parchment obeys the differential equation y -ky, where k is a positive constant. The decay law calculates the number of undecayed nuclei in a given radioactive substance. We start by noting that the speed of radioactive decays occurring in a sample is proportional to the number of radioactive atoms in the sample. Half Life explained with interactive graphics, examples, practice problems, and real world examples of radioactive. A quantity is subject to exponential decay if it decreases at a rate proportional to its current value. WARNING: there is a little bit of calculus involved. There are many general forms of the equation that deal with chains of nuclides, but here we only worry about the basics. This equation allows us to figure out how many radioactive atoms are left after any amount of time. To find the percent of Carbon 14 remaining after a given number of years, type in the number of years and click on Calculate. This page derives the basic equation of radioactive decay. The Math behind Radioactive Decay By Nick Touran, Ph.D.,
